


Wellion INOVO FIA Analyzer
SARS-COV-2 ANTIBODY DETERMINATION
Reliable test result in BAU/mL with only one finger prick in 15 min!
Reliable test result in BAU/mL with only one finger prick in 15 min!
The Wellion INOVO FIA Analyzer in compact benchtop format allows a fast and easy SARS-CoV-2 antibody determination. With only one finger prick (20µl capillary blood) the evaluation can be done directly on site in only 15 minutes.
- Quick
- User-friendly - easy operation
- Compact benchtop format
- Easy on-site determination
- Large, clear touch screen
- Integrated thermal printer - results can be taken away immediately
- 3 different test options, depending on need and frequency
- Standard
- Quick
- Serial test
SARS-CoV-2 antibody detection - The reliable test result to take home
- Checking the immune response
- Determination of SARS-CoV-2 S antibodies (spike antibodies*)
- Result in only 15 minutes
- No waiting time - evaluation right on the spot
- Small amount of blood (20 µl)
- Convenient measurement with capillary blood (finger prick with safety lancet)
- Quantitative information in BAU/mL

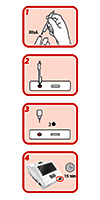
See how to perform a measurement here: https://youtu.be/2nhZ1U30EdM

Scheme of the different test procedures for the SARS-CoV-2 antibody test with the Wellion INOVO FIA Analyzer
- Test preparation
- Incubation and measurement
Instant - timecode 04:51
Batch - timecode 07:30
Specifications
- Measurement of SARS-CoV-2 S antibodies*
- Quantitative result in BAU/mL (= WHO standard)
- Reliable - performance data for whole blood from finger prick:
- Sensitivity: 98,22%
- Specificity: >99,99%
- Accuracy: 99,39%
- tests/kit
- 24 months shelf life
- Storage at room temperature
- Calibration by code chip (included in test kit)
*Spike (S)-glycoprotein is the main target of neutralisation by SARS-CoV-2 antibodies and thus the basis for vaccine development.
Approved for professional use only.

